Before we begin tonight, please join me in paying my respects to my mum, who would have been 91 years old today. The season beginning with Thanksgiving and lasting through New Year’s Day was her favorite of the year, and she showed lots of love to everyone during this time. But she showed lots of love all year ’round.
The definition of an emulsion is two dissimilar liquids that are dispersed into a more of less long lasting mixture that has properties different than either of the two liquids. I say dissimilar because in most cases one of the liquids is hydrophopic (literally, “water fearing”, often an oil or hydrocarbon) and the other one hydrophilic (literally, “water loving”, often water itself).
The old adage that oil and water do not mix is only partially true. It is possible to make them mix, and it is often done intentionally. Sometimes it happens upon accident, and we organic chemists know that when the synthetic product that we seek to isolate forms an emulsion with the solvent and/or other materials in the separatory funnel that is easy to become piqued by that.
An emulsion is a special case of the more general suspension, where one material is dispersed in another. Solutions are different than suspensions because in solutions the solute (dissolved material) is in the form of single molecules or ions, where in a suspension the divided phase is much larger than on the molecular level, actually being aggregates of many molecules.
Not only two liquids can form suspensions. Smoke is a suspension of a solid in a gas, whilst whipped cream is a suspension of a gas in a liquid. Suspensions can occur betwixt any two normal phases of matter except for gas in gas mixtures which, since they are at the molecular level, are true solutions.
When you mix oil and vinegar the droplets of oil quickly gather together into larger droplets and finally rise to the top, forming a separate layer. Regardless of how much you stir or blend, a stable suspension will not form. The smaller the oil droplets are the longer they will stay in the water, but they eventually separate into a distinct layer. We shall get back to overcoming that later.
Milk in a suspension of butterfat (the divided phase) in water (the continuous phase). Actually, milk will separate in time (cream rises), but the separation is not complete unless centrifugation is employed. Before the invention of the cream separator all milk had at least some fat in it, and cream was not as concentrated in fat as it can be now. This is because there are some minor components in milk that facilitate forming a stable emulsion. Whole milk (legally, at least 3.25% butterfat) will separate into two layers unless it is homogenized. This involves subjecting the milk to vigorous mechanical action with the purpose of reducing the size of the butterfat particles. This allows the minor components to be more effective in keeping the butterfat globules in suspension. Milk, at least from the refrigerator, is technically not an emulsion because butterfat is a solid at that temperature. However, at body temperature the butterfat melts, so warm milk is a true emulsion.
The minor components in milk that facilitate making a stable emulsion are called, as you may have guessed, emulsifying agents. In general, emulsifying agents have some soap like properties in that they have a small, hydrophilic region and a long, hydrophobic region. The hydrophilic region is strongly attracted to water whilst the hydrophilic region is strongly attracted to oils and fats, or other nonpolar molecules, if we confine our discussion to water based emulsions.
Let us take our oil and vinegar again, but first let us add an egg, or an egg yolk, to the vinegar before we add most of the oil. Let us take, say 30 mL (2 tablespoons) of vineger (or lemon juice, or half and half), one egg yolk, and 15 mL of oil to start. Then we stir, stir (or use a blender or food processor) and add more oil little by little with lots of agitation the whole time. If we keep going, we will find that we can add as much as 500 mL of oil to the vinegar and it does not separate. As a matter of fact, we just made mayonnaise!
This stable emulsion has properties quite different from either the oil or the vinegar. First of all, it is opaque whereas both the oil and the vinegar is transparent. This is because of the billions and billions of oil droplets suspended in the continuous phase (the vinegar). These droplets are of such a size that light is scattered by them rather than passing through. Second, the consistency of the emulsion is much thicker than either the oil or the vinegar because of the friction betwixt the billions of oil droplets and the water tends to reduce ease of motion.
The emulsifying agent (or just emulsifier) in the egg yolk is primarily lecithin. Here is a structural formula for a typical lecithin molecule (other fatty acids are also present, since natural lecithin is a mixture of several variants).
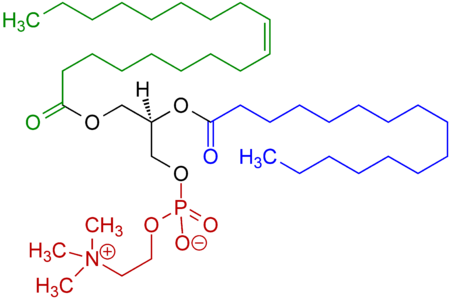
The red region is the small, hydrophilic part of the molecule whilst the blue and green parts are the hydrophobic region.
Actually, mayonnaise made with only these ingredients will slowly separate. This did not matter much before refrigeration, when mayonnaise was made for immediate consumption. With refrigeration and preservatives, mayonnaise lasts for a long time so stabilizers are added to make the emulsion more long lasting. Proteins make pretty good stabilizers, so often whole eggs are used to make it, and ground mustard seeds are often added because they contain a gum that acts as a stabilizer and also the seeds add flavor.
If not gotten too hot or frozen, properly stabilized mayonnaise will not separate until it goes bad. Heat denatures the protein stabilizers and causes separation, and freezing solidifies both phases which collapse when thawed back to two separate ones.
There are lots of other emulsions both in culinary and industrial practice. Water based paint is an emulsion with water as the continuous phase and synthetic polymers as the discontinuous phase. Solids are also added to such paint, and they are not suspended so that is why it has to be stirred or shaken before use. I used to work in a mom and pop paint factory and we added quite a little lecithin from soy to our water based paint to emulsify it.
When I was in graduate school I sometimes had to do some synthetic organic chemistry to produce materials for my photochemical research. I have never claimed to be a great synthetic chemist, but I could follow recipes fairly well. Sometimes when it came time to work up a reaction by adding water, and emulsion would form. Trying to work with an emulsion whilst purifying products is extremely difficult, because the desired material is pretty much isolated from whatever solvent you are trying to use to extract it. Sometimes I could break the emulsion by adding a little methanol, and sometimes I could take it to the biochemistry section and use a high speed centrifuge to break them. But I always hated it when I got an emulsion because they were trouble.
In cooking, breaking an emulsion is usually not the goal, but rather to form a stable one. When making the wine (or vinegar) and butter sauce beurre blanc the goal is to form an emulsion of butterfat and the discontinuous phase and (primarily) water as the continuous phase. Since the sauce is at a temperature above the melting point of butterfat, it is a true emulsion. If you get it too hot, over around 135 degrees F, it will break. You can sometimes restore it by adding a splash of cold water or wine and whisking it, and sometimes you can not. Letting it get too cold will break it as well, because when the butterfat solidifies the sharp (on the microscopic level) crystals of butterfat penetrate the emulsifier membranes and glom onto each other, and it separates when reheated. If that happens you have to start over, but you can add some of the broken sauce to the new one if you do not overdo it.
Emulsions are used frequently in medicine because lots of drugs to not dissolve in water. An example is the drug Propofol, best known for being the agent that caused Michael Jackson’s death. Propofol is an excellent intervenous anesthetic agent, made of 1% 1,6-diisopropyl phenol (a liquid at room temperature that is insoluble in water), 10% soy oil, 1.2% egg lecithin, some glycerol, and the rest water. The lecithin emulsifies the active drug and oil into the water, forming a stable emulsion so that it can be given IV.
Probably the most common emulsion encountered is something about which you never think, and you handle it every day. It is an oil in water emulsion that is formed when soap or detergent and water are applied to the skin, to soiled clothing, or to dirty dishes. The soap or detergent is the emulsifying agent, and the resulting oil in water emulsion is easily rinsed off of the item being cleaned. Skin and clothes have lots of oil on them from the sebaceous glands, and without soap or detergent it is really hard to rinse away from them. But add a little soap and the situation changes. Some fats are solids at room temperature, so dirty dishes are a bit different than skin or clothes. This is why hot water is helpful. It is much harder to make a solid in water dispersion than a liquid in water one because of kinetics: it takes longer to make a dispersion of a solid in water because the attack rate of the emulsifier is slower. There is also a purely mechanical reason, too. The melted fat is dislodged from the item faster just because it the liquid is more mobile.
That does it for tonight. Please keep those comments, questions, and especially any corrections coming. I always learn more than I could possibly hope to teach in writing this series. Tips and recs are also always welcome. I plan to be here for most of the evening, so should be able to field any comments in near real time. Tomorrow evening the plan is to come back at around 9:00 Eastern for any late comments.
Warmest regards,
Doc, aka Dr. David W. Smith
Crossposted at
Docudharma, and

1 comment
Author
mixing things up well?
Warmest regards,
Doc